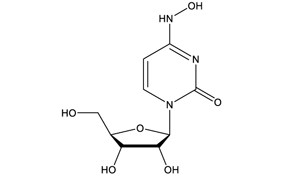
N4-Hydroxycytidine | Novel antiviral
NMR (Conforms)
Available Options
| Size: | Price | Quantity | |
|---|---|---|---|
| 5 mg | $70.00 | ||
| 25 mg | $250.00 |
N4-Hydroxycytidine (3258-02-4) was originally identified as a mutagen effecting AT to GC base-pair transitions.1 It has also been found to have antiviral properties against a broad range of viruses including hepatitis C2, norovirus3, Ebola virus4, Chikungunya virus5, influenza and respiratory syncytial viruses6, and importantly, coronaviruses.7,8 N4-hydroxycytidine is the active molecule in the antiviral pro-drug clinical candidate EIDD-2801.9
References/Citations:
1) Janion and Glickman (1980), N4-hydroxycytidine: a mutagen specific for AT to GC transitions; Mutat. Res., 72 43
2) Stuyver et al. (2003), Ribonucleotide analogue that blocks replication of bovine viral diarrhea and hepatitis C viruses in culture; Antimicrob. Agents Chemother., 47 244
3) Costantini et al. (2012), Antiviral activity of nucleoside analogues against norovirus; Antivir. Ther., 17 981
4) Reynard et al. (2015), Identification of a New Ribonucleoside Inhibitor of Ebola Virus Replication; Viruses, 7 6233
5) Ehteshami et al. (2017), Characterization of ß-D-N4-Hydroxycytidine as a Novel Inhibitor of Chikungunya Virus; Antimicrob. Agents Chemother., 61 e02395-16
6) Yoon et al. (2018), Orally efficacious broad-spectrum ribonucleoside analog inhibitor of influenza and respiratory syncytial viruses; Antimicrob. Agents Chemother., 62 e00766-18
7) Barnard et al. (2004), Inhibition of severe acute respiratory syndrome-associated coronavirus (SARSCoV) by calpain inhibitors and beta-D-N4-hydroxycytidine; Antivir. Chem. Chemother., 15 15
8) Pyrc et al. (2006), Inhibition of human coronavirus NL63 infection at early stages of the replication cycle; Antimicrob. Agents Chemother., 50 2000
9) Toots et al. (2019), Characterization of orally efficacious influenza drug with high resistance barrier in ferrets and human airway epithelia; Sci. Transl. Med., 11 eaax5866
NMR (Conforms)
Safety Data Sheet:
Product Data Sheet:
Materials provided by Focus Biomolecules are for laboratory research use only and are not intended for human or veterinary applications. Please note that we do not sell to individuals and that all orders placed by non-research organizations will incur a $20 restocking/refund fee
N4-Hydroxycytidine (3258-02-4) was originally identified as a mutagen effecting AT to GC base-pair transitions.1 It has also been found to have antiviral properties against a broad range of viruses including hepatitis C2, norovirus3, Ebola virus4, Chikungunya virus5, influenza and respiratory syncytial viruses6, and importantly, coronaviruses.7,8 N4-hydroxycytidine is the active molecule in the antiviral pro-drug clinical candidate EIDD-2801.9
References/Citations:
1) Janion and Glickman (1980), N4-hydroxycytidine: a mutagen specific for AT to GC transitions; Mutat. Res., 72 43
2) Stuyver et al. (2003), Ribonucleotide analogue that blocks replication of bovine viral diarrhea and hepatitis C viruses in culture; Antimicrob. Agents Chemother., 47 244
3) Costantini et al. (2012), Antiviral activity of nucleoside analogues against norovirus; Antivir. Ther., 17 981
4) Reynard et al. (2015), Identification of a New Ribonucleoside Inhibitor of Ebola Virus Replication; Viruses, 7 6233
5) Ehteshami et al. (2017), Characterization of ß-D-N4-Hydroxycytidine as a Novel Inhibitor of Chikungunya Virus; Antimicrob. Agents Chemother., 61 e02395-16
6) Yoon et al. (2018), Orally efficacious broad-spectrum ribonucleoside analog inhibitor of influenza and respiratory syncytial viruses; Antimicrob. Agents Chemother., 62 e00766-18
7) Barnard et al. (2004), Inhibition of severe acute respiratory syndrome-associated coronavirus (SARSCoV) by calpain inhibitors and beta-D-N4-hydroxycytidine; Antivir. Chem. Chemother., 15 15
8) Pyrc et al. (2006), Inhibition of human coronavirus NL63 infection at early stages of the replication cycle; Antimicrob. Agents Chemother., 50 2000
9) Toots et al. (2019), Characterization of orally efficacious influenza drug with high resistance barrier in ferrets and human airway epithelia; Sci. Transl. Med., 11 eaax5866
Calculate the molar concentration, mass or volume in a solution.
Concentration × Volume × Molecular Weight = Mass
Focus Biomolecules • Plymouth Meeting, PA USA • 1-855-FOCUS21
Focus Biomolecules
Plymouth Meeting, PA USA
1-855-FOCUS21
Website Created by Advanta Advertising LLC.

