Abemaciclib | Potent and selective CDK 4/6 inhibitor
NMR (Conforms)
Available Options
| Size: | Price | Quantity | |
|---|---|---|---|
| 10 mg | $50.00 | ||
| 50 mg | $115.00 |
Abemaciclib (1231930-82-7) is a potent and selective CDK4/6 inhibitor (IC50 = 2 nM and 10 nM respectively).1 It caused G1 cell cycle arrest in colo-205 colorectal cells, MDA-MB-361 breast cancer cells, and MV4-11 AML cells. Abemaciclib was also active in several human tumor xenograft models. It displayed efficacy in patients with various solid tumors including breast cancer, non-small cell lung cancer, glioblastoma, melanoma, colorectal cancer, and hormone receptor-positive breast cancer.2 Abemaciclib induced a T cell inflamed tumor microenvironment and enhanced the efficacy of PD-L1 checkpoint blockade in MCF-7 breast cancer cells.3 FDA approved for the treatment of advanced breast cancers.
References/Citations:
1) Gelbert et al. (2014), Preclinical characterization of the CDK4/6 inhibitor LY2835219: In-vivo cell cycle-dependent/independent anti-tumor activities alone/in combination with gemcitabine; Invest. New Drugs, 32 825
2) Patnaik et al. (2016), Efficacy and Safety of Abemaciclib, an Inhibitor of CDK4 and CDK6, for Patients with Breast Cancer, Non-small Cell Lung Cancer, and Other Solid Tumors; Cancer Discov., 6 740
3) Schaer et al. (2018), The CDK4/6 Inhibitor Abemaciclib Induces a T Cell Inflamed Tumor Microenvironment and Enhances the Efficacy of PD-L1 Checkpoint Blockade; Cell Rep., 22 2978
NMR (Conforms)
Safety Data Sheet:
Product Data Sheet:
Materials provided by Focus Biomolecules are for laboratory research use only and are not intended for human or veterinary applications. Please note that we do not sell to individuals and that all orders placed by non-research organizations will incur a $20 restocking/refund fee
Abemaciclib (1231930-82-7) is a potent and selective CDK4/6 inhibitor (IC50 = 2 nM and 10 nM respectively).1 It caused G1 cell cycle arrest in colo-205 colorectal cells, MDA-MB-361 breast cancer cells, and MV4-11 AML cells. Abemaciclib was also active in several human tumor xenograft models. It displayed efficacy in patients with various solid tumors including breast cancer, non-small cell lung cancer, glioblastoma, melanoma, colorectal cancer, and hormone receptor-positive breast cancer.2 Abemaciclib induced a T cell inflamed tumor microenvironment and enhanced the efficacy of PD-L1 checkpoint blockade in MCF-7 breast cancer cells.3 FDA approved for the treatment of advanced breast cancers.
References/Citations:
1) Gelbert et al. (2014), Preclinical characterization of the CDK4/6 inhibitor LY2835219: In-vivo cell cycle-dependent/independent anti-tumor activities alone/in combination with gemcitabine; Invest. New Drugs, 32 825
2) Patnaik et al. (2016), Efficacy and Safety of Abemaciclib, an Inhibitor of CDK4 and CDK6, for Patients with Breast Cancer, Non-small Cell Lung Cancer, and Other Solid Tumors; Cancer Discov., 6 740
3) Schaer et al. (2018), The CDK4/6 Inhibitor Abemaciclib Induces a T Cell Inflamed Tumor Microenvironment and Enhances the Efficacy of PD-L1 Checkpoint Blockade; Cell Rep., 22 2978
Related products
-
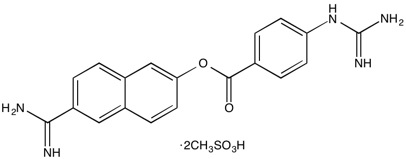
Nafamostat Mesylate | Serine protease inhibitor
From $50.00CAS:82956-11-4Catalog Number:10-1089Activity:Serine protease inhibitorAlternate Names:Futhan; FUT-175Chemical Name:6-Amidino-2-naphthyl-4-guanidinobenzoate dimesylateMolecular Weight:539.59Molecular Formula:C19H17N5O2·2CH4O3SSolubility:Soluble in Water (up to 25 mg/ml).Physical Properties:White solidPurity:98% by TLC
NMR (Conforms)Storage Temperature:RTStability:Stable for 2 years from date of purchase as supplied. Solutions in distilled water may be stored at 4°C for up to 1 month, or at -80°C for up to 6 months.Shipping Code:RT -
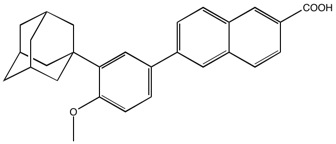
Adapalene | Retinoid RARbeta, gamma agonist
From $30.00CAS:106685-40-9Catalog Number:10-1011Activity:Retinoid RARβ,γ agonistAlternate Names:CD271Chemical Name:6-[3-(1-Adamantyl)-4-methoxyphenyl]-2-naphthoic acidMolecular Weight:412.53Molecular Formula:C28H28O3Solubility:Soluble in DMSO (up to 3 mg/ml) or in DMF (up to 6 mg/ml).Physical Properties:White solidPurity:98% by TLC
NMR (Conforms)Storage Temperature:RTStability:Stable for 1 year from date of purchase as supplied. Solutions in DMSO or DMF may be stored at -20°C for up to 3 monthsShipping Code:RT -
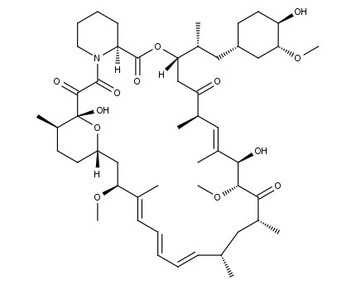
Rapamycin | mTOR inhibitor
From $52.00CAS:53123-88-9Catalog Number:10-1104Activity:mTOR inhibitorAlternate Names:SirolimusMolecular Weight:914.17Molecular Formula:C51H79NO13Solubility:Soluble in DMSO (up to 40 mg/ml) or in Ethanol (up to 90 mg/ml).Physical Properties:White solidPurity:98%
NMR (Conforms)Storage Temperature:-20°CStability:Stable for 2 years from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20°C for up to 2 months.Shipping Code:RT
Calculate the molar concentration, mass or volume in a solution.
Concentration × Volume × Molecular Weight = Mass
Focus Biomolecules • Plymouth Meeting, PA USA • 1-855-FOCUS21
Focus Biomolecules
Plymouth Meeting, PA USA
1-855-FOCUS21
Website Created by Advanta Advertising LLC.