ALLN | Calpain inhibitor
NMR (Conforms)
Available Options
| Size : | Price | Quantity | |
|---|---|---|---|
| 5 mg | $50.00 | ||
| 25 mg | $188.00 |
ALLN (110044-82-1) is a cell-permeable, peptide aldehyde inhibitor of calpain I (Ki=190 nM), calpain II (Ki=150 nM), cathepsin L (Ki=0.5 nM) and other neutral cysteine proteases.1 Inhibits cell cycle progression at G1/S and metaphase/anaphase in CHO cells by inhibiting cyclin B degradation.2 Inhibits proteolytic degradation of IκBα and IκBβ in RAW macrophages induced with LPS.3 Blocks AIF-mediated necroptosis.4 Cell permeable.
References/Citations:
1) Sasaki et al. (1990), Inhibitory effect of di- and tripeptidyl aldehydes on calpains and cathepsins; J. Enzyme Inhib. 3 195
2) Sherwood et al. (1993) In vivo inhibition of cyclin B degradation and induction of cell-cycle arrest in mammalian cells by the neutral cysteine protease inhibitor N-acetylleucylleucylnorleucinal; Proc. Natl. Acad. Sci USA 90 3353
3) Schow and Joly (1997), N-acetyl-leucinyl-leucinyl-norleucinal inhibits lipopolysaccharide-induced NF-kappaB activation and prevents TNF and IL-6 synthesis in vivo; Cell Immunol. 175 199
4) Shang et al. (2014) Calpain: a molecule to induce AIF-mediated necroptosis in RGC-5 following elevated hydrostatic pressure; BMC Neurosci. 15 63
NMR (Conforms)
Safety Data Sheet:
Product Data Sheet:
Materials provided by Focus Biomolecules are for laboratory research use only and are not intended for human or veterinary applications. Please note that we do not sell to individuals and that all orders placed by non-research organizations will incur a $20 restocking/refund fee
ALLN (110044-82-1) is a cell-permeable, peptide aldehyde inhibitor of calpain I (Ki=190 nM), calpain II (Ki=150 nM), cathepsin L (Ki=0.5 nM) and other neutral cysteine proteases.1 Inhibits cell cycle progression at G1/S and metaphase/anaphase in CHO cells by inhibiting cyclin B degradation.2 Inhibits proteolytic degradation of IκBα and IκBβ in RAW macrophages induced with LPS.3 Blocks AIF-mediated necroptosis.4 Cell permeable.
References/Citations:
1) Sasaki et al. (1990), Inhibitory effect of di- and tripeptidyl aldehydes on calpains and cathepsins; J. Enzyme Inhib. 3 195
2) Sherwood et al. (1993) In vivo inhibition of cyclin B degradation and induction of cell-cycle arrest in mammalian cells by the neutral cysteine protease inhibitor N-acetylleucylleucylnorleucinal; Proc. Natl. Acad. Sci USA 90 3353
3) Schow and Joly (1997), N-acetyl-leucinyl-leucinyl-norleucinal inhibits lipopolysaccharide-induced NF-kappaB activation and prevents TNF and IL-6 synthesis in vivo; Cell Immunol. 175 199
4) Shang et al. (2014) Calpain: a molecule to induce AIF-mediated necroptosis in RGC-5 following elevated hydrostatic pressure; BMC Neurosci. 15 63
Related products
-
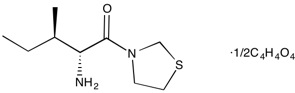
P32/98 | DPPIV inhibitor
From $100.00CAS:136259-20-6Catalog Number:10-1142Activity:DPPIV inhibitorAlternate Names:Isoleucine thiazolidide hemi-fumarateChemical Name:(2S,3S)-2-Amino-3-methyl-1-(thiazolidin-3-yl)pentan-1-one hemi-fumarateMolecular Weight:260.36Molecular Formula:C9H18N2OS·1/2(C4H4O4)Solubility:Soluble in DMSO (up to 25 mg/ml) or in Water (up to 20 mg/ml).Physical Properties:White solidPurity:98% by TLC
NMR (Conforms)Storage Temperature:-20°CStability:Stable for 2 years from date of purchase as supplied. Solutions in DMSO or distilled water may be stored at -20°C for up to 1 month.Shipping Code:RT -
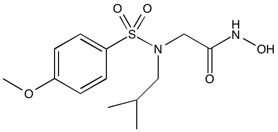
NNGH | MMP inhibitor (pan specific)
From $55.00CAS:161314-17-6Catalog Number:10-1045Activity:MMP inhibitor (pan spec.)Chemical Name:N-Isobutyl-N-(4-methoxyphenylsulfonyl)glycyl hydroxamic acidMolecular Weight:316.38Molecular Formula:C13H20N2O5SSolubility:Soluble in DMSO (up to 30 mg/ml) or in Ethanol (up to 8 mg/ml).Physical Properties:White solidPurity:98% by TLC
NMR (Conforms)Storage Temperature:RTStability:Stable for 1 year from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20°C for up to 3 months.Shipping Code:RT -
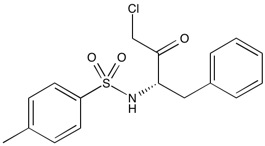
TPCK | Serine protease inhibitor
From $32.00CAS:402-71-1Catalog Number:10-2290Activity:Serine protease inhibitorChemical Name:N-p-Tosyl-L-phenylalanine chloromethyl ketoneMolecular Weight:351.8Molecular Formula:C17H18ClNO3SSolubility:Soluble in DMSO (up to 10 mg/ml).Physical Properties:White solidPurity:98% by TLC
NMR (Conforms)Storage Temperature:-20°Stability:Stable for 1 year from date of purchase as supplied. Solutions in DMSO may be stored at -20° for up to 3 months.Shipping Code:RT
Calculate the molar concentration, mass or volume in a solution.
Concentration × Volume × Molecular Weight = Mass
Focus Biomolecules • Plymouth Meeting, PA USA • 1-855-FOCUS21
Focus Biomolecules
Plymouth Meeting, PA USA
1-855-FOCUS21
Website Created by Advanta Advertising LLC.