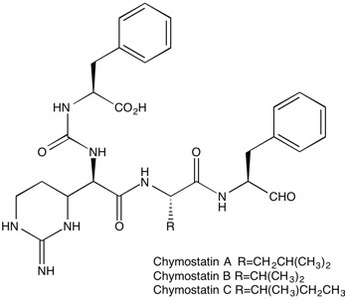
Chymostatin | Inhibitor of Chymotrypsin-like serine proteases
NMR (Conforms)
Available Options
| Size : | Price | Quantity | |
|---|---|---|---|
| 5 mg | $98.00 | ||
| 25 mg | $400.00 |
Chymostatin (9076-44-2) is a potent, competitive, slow-binding inhibitor of a-, b-, g-, d-chymotrypsin, papain and cathepsins B/G (chymotryptase-like serine proteases)1, Ki=9.36 and 13.1 nM for chymotrypsin and chymase2. Cathepsin G Ki=0.15 mM.3 Consists of a mixture of type A (L-Leu), B (L-Val) and C (L-Ile) forms.1 Decreases plasma and tissue levels of angiotensin II without lowering mean blood pressure in a hypertensive rat model.4 Commonly used in lysis buffers to prevent degradation of proteins. Typical working concentration is 6-60 µg/ml.
References/Citations:
1) Umezawa et al. (1970), Chymostatin, a new chymotrypsin inhibitor produced by actinomycetes; J. Antibiot. (Tokyo) 23 425
2) Johnson et al. (1998), Inactivation of chymotrypsin and human skin chymase: kinetics of time-dependent inhibition in the presence of substrate; Biochim. Biophys. Acta 953 269
3) Stein and Strimpler (1987), Slow-binding of chymotrypsin and cathepsin G by the peptide aldehyde chymostatin; Biochemistry 26 2611
4) Roszkowska-Chojecka et al. (2015), Effects of chymostatin, a chymase inhibitor, on blood pressure, plasma and tissue angiotensin II, renal haemodynamics and renal excretion in two models of hypertension in the rat; Exp. Physiol. 100 1093
NMR (Conforms)
Safety Data Sheet:
Product Data Sheet:
Materials provided by Focus Biomolecules are for laboratory research use only and are not intended for human or veterinary applications. Please note that we do not sell to individuals and that all orders placed by non-research organizations will incur a $20 restocking/refund fee
Chymostatin (9076-44-2) is a potent, competitive, slow-binding inhibitor of a-, b-, g-, d-chymotrypsin, papain and cathepsins B/G (chymotryptase-like serine proteases)1, Ki=9.36 and 13.1 nM for chymotrypsin and chymase2. Cathepsin G Ki=0.15 mM.3 Consists of a mixture of type A (L-Leu), B (L-Val) and C (L-Ile) forms.1 Decreases plasma and tissue levels of angiotensin II without lowering mean blood pressure in a hypertensive rat model.4 Commonly used in lysis buffers to prevent degradation of proteins. Typical working concentration is 6-60 µg/ml.
References/Citations:
1) Umezawa et al. (1970), Chymostatin, a new chymotrypsin inhibitor produced by actinomycetes; J. Antibiot. (Tokyo) 23 425
2) Johnson et al. (1998), Inactivation of chymotrypsin and human skin chymase: kinetics of time-dependent inhibition in the presence of substrate; Biochim. Biophys. Acta 953 269
3) Stein and Strimpler (1987), Slow-binding of chymotrypsin and cathepsin G by the peptide aldehyde chymostatin; Biochemistry 26 2611
4) Roszkowska-Chojecka et al. (2015), Effects of chymostatin, a chymase inhibitor, on blood pressure, plasma and tissue angiotensin II, renal haemodynamics and renal excretion in two models of hypertension in the rat; Exp. Physiol. 100 1093
Related products
-
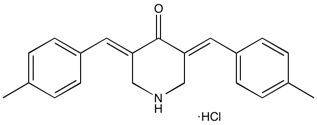
NSC632839 | DUB inhibitor
From $65.00CAS:157654-67-6Catalog Number:10-1385Activity:DUB inhibitorAlternate Names:F6Chemical Name:3,5-Bis[(4-methylphenyl)methylene]-4-piperidone hydrochlorideMolecular Weight:339.86Molecular Formula:C21H21NO HClSolubility:Soluble in DMSO (up to 8 mg/ml).Physical Properties:Yellow solidPurity:98% by HPLC
NMR (Conforms)Storage Temperature:-20°CStability:Stable for 2 years from date of purchase as supplied. Solutions in DMSO may be stored at -20° for up to 1 month.Shipping Code:RT -
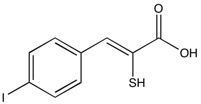
PD-150606 | Calpain Inhibitor
From $69.00CAS:179528-45-1Catalog Number:10-1515Activity:Calpain InhibitorChemical Name:(Z)-3-(4-Iodophenyl)-2-mercapto-2-propenoic acidMolecular Weight:306.12Molecular Formula:C9H7IO2SSolubility:Soluble in DMSO (up to 30 mg/ml) or in Ethanol (up to 15 mg/ml).Physical Properties:Yellow solidPurity:98% by TLC
NMR (Conforms)Storage Temperature:-20°C (des.)Stability:Stable for 2 years from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20° for up to 3 months.Shipping Code:RT -
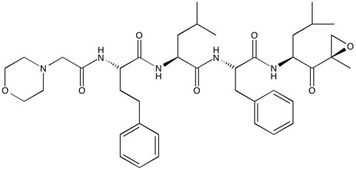
Carfilzomib | Proteasome inhibitor
From $50.00CAS:868540-17-4Catalog Number:10-1477Activity:Proteasome inhibitorAlternate Names:PR-171Chemical Name:(αS)-α-[[2-(4-morpholinyl)acetyl]amino]benzenebutanoyl-L-leucyl-N-[(1S)-3-methyl-1-[[(2R)-2-methyl-2-oxiranyl]carbonyl]butyl]- L-PhenylalaninamideMolecular Weight:719.93Molecular Formula:C40H57N5O7Solubility:Soluble in DMSO (up to 80 mg/ml) or in Ethanol (up to 25 mg/ml).Physical Properties:White solidPurity:98% by TLC
NMR (Conforms)Storage Temperature:-20°Stability:Stable for 1 year from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20° for up to 1 week.Shipping Code:RT
Calculate the molar concentration, mass or volume in a solution.
Concentration × Volume × Molecular Weight = Mass
Focus Biomolecules • Plymouth Meeting, PA USA • 1-855-FOCUS21
Focus Biomolecules
Plymouth Meeting, PA USA
1-855-FOCUS21
Website Created by Advanta Advertising LLC.